Glomerular Filtration Rate (FIT-GFR) Test
Technical Information
Drug safety has always played a key role in drug development. Unfortunately, drug toxicity studies often omit careful assessment of kidney function as indicated by recent publications.
Accurate measurements of kidney function using a measured glomerular filtration rate (GFR) test provides necessary and reliable information, while biomarker-based estimates are often misleading. On May 2010 (1), the FDA published a draft recommending that the drug development process should include a "measure" GFR.
Based on functional immunoassay technology (FIT™), BioPAL has developed immunoassay kits to measure the renal clearance of ideal filtration markers in order to obtain an accurate GFR measurement. We offer two different kits - Inulin (the gold standard) and Iohexol. Gadolinium-based kits have been discontinued (Gd-DTPA and Gd-DOTA).
(1) A recommendation from the conference is that in drug development, GFR should be measured directly with an inert tracer (e.g. inulin, iohexol, iothalamate, etc.) to determine pharmacokinetic or pharmacodynamic alterations due to kidney dysfunction. FDA KDIGO. Released draft. Guidance for Industry: Pharmacokinetics in Patients with Impaired Renal Function - Study Design, Data Analysis, and Impact on Dosing and Labeling. Docket Number FDA-2010-D-0133 May 16, 2010.
FOR RESEARCH USE ONLY.
NOT FOR USE IN DIAGNOSTIC PROCEDURES.
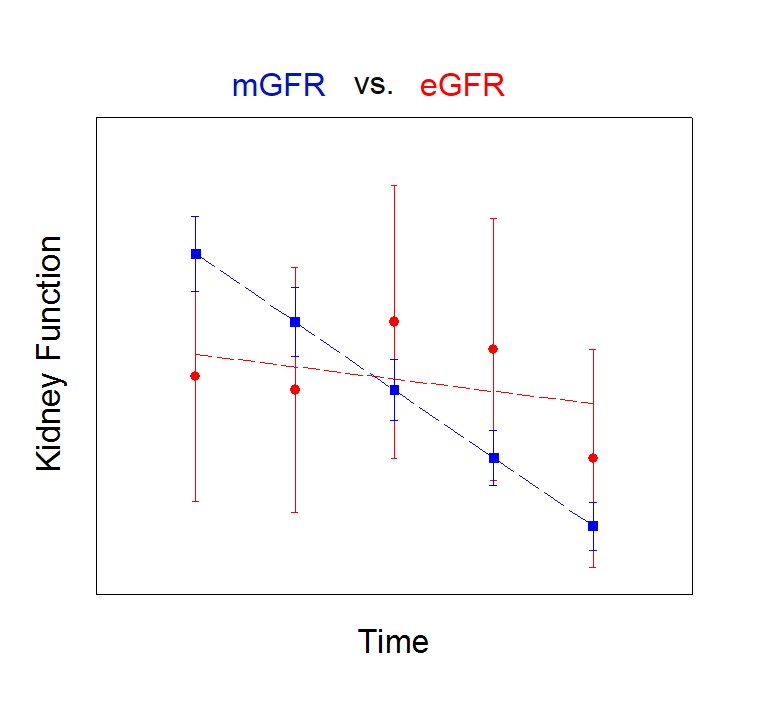
Due to the error associated with estimating GFR (eGFR) using biomarkers and correction equations, the above figure illustrates that researchers may miss important trends using eGFR that would otherwise be obvious using a direct measurement of kidney function (mGFR).
USP Inulin
BioPAL’s Inulin for Injection (USP) is for preclinical applications ONLY. However, BioPAL offers USP Inulin (I-1000) for researchers need to measure inulin clearance for human clinical research. Researchers may purchase USP Inulin (Solid) as a dry powder and then utilize a compounding pharmacy for formulation. BioPAL’s Inulin ELISA kits (FIT-0415) can then be used to measure its concentration is collected samples.